
By highlighting that much has been achieved but that there is still much to do this review aims to inspire, focus and guide future efforts in this area. We focus on complexes whose identity has been unambiguously established by structural authentication by single-crystal X-ray diffraction with respect to their synthesis, characterisation, bonding, and reactivity, in order to provide a representative overview of this burgeoning area. It is a reactive nonmetal and is the 13 th most abundant element on earth. The comparison will provide useful information on different photochemical pathways of d- and f-element complexes. It is a pale yellow or green gas, whose name comes from the Latin word fluere, which means to flow. Slow relaxation in f-elements The electronic structure of f-element compounds is profoundly dierent to that of d-block elements. University: North Central Missouri College. Explain why noble gases are inert and do not form ions. They are placed in two separate rows at the bottom of the period table. Fluorine (F) is a chemical element of the periodic table, located in the group 17 and the period 2, and is having the atomic number 9. Provide one element as an example to support your reasoning. They belong to the f-block of the periodic table. F-elements are called inner transition elements. As the most electronegative reactive element, it is extremely reactive, as it reacts with all other elements except for the light inert gases. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Here, we review complexes containing chemical bonds between the f-elements and heavy pnictogens from phosphorus to bismuth that spans five decades of endeavour. What is the difference between a transition metal and an f-element Transition metals are in the d-block and have valance electrons in the d-orbital. Fluorine is a chemical element it has symbol F and atomic number 9. In contrast, the corresponding f-element chemisty with the heavier pnictogen analogues phosphorus, arsenic, antimony, and bismuth has remained significantly underdeveloped, due largely to a lack of suitable synthetic methodologies and also the inherent hard(f-element)–soft(heavier pnictogen) acid–base mismatch, but has begun to flourish in recent years. This periodic table chart lists elements by name in alphabetical order including the element. amides, imides, and nitrides, has become well developed over many decades. A silicon donor atom is commonly introduced to an. 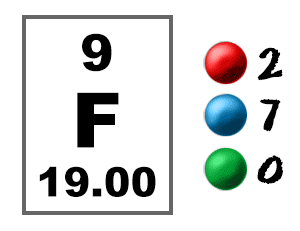
The coordination and organometallic chemistry of the f-elements, that is group 3, lanthanide, and actinide ions, supported by nitrogen ligands, e.g. The increasing rate of development in f-element silicon chemistry in the last two decades has provided the motivation for this perspective the heavier tetrels are discussed herein to inspire the future development of the f-element chemistry of these elements also, but silicon remains the focus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
